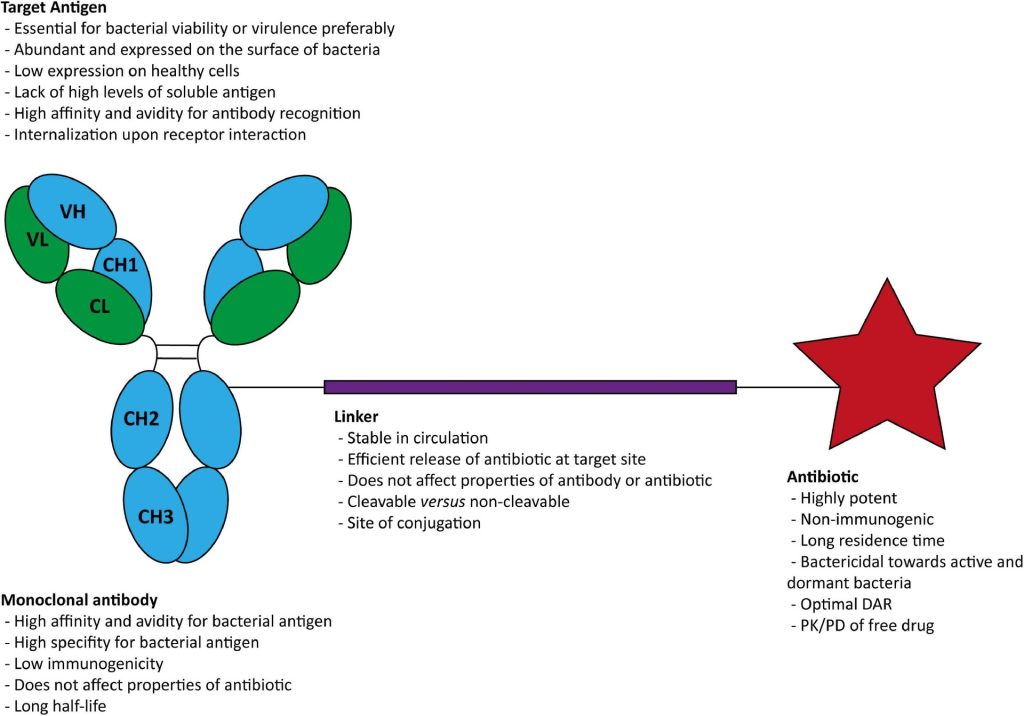
The rapid rise of antimicrobial resistance (AMR) presents a critical challenge in modern healthcare. Traditional antibiotics are becoming increasingly ineffective against resistant strains of bacteria, necessitating innovative therapeutic strategies. One promising approach is the development of antibody-antibiotic conjugates (AACs). These innovative agents use the selectivity, favorable pharmacokinetics (PK), and safety profile of antibodies, enabling the administration of more potent antibiotics with fewer off-target effects which combine the specificity of antibodies with the bactericidal properties of antibiotics to target and eliminate bacterial infections with high precision. Although AAC development is challenging due to the complexity of the antibody, antibiotic, and linker components, several examples are currently under development.1
Mechanism of Action
AACs leverage the targeted binding capabilities of monoclonal antibodies (mAbs) to deliver potent antibiotics directly to bacterial cells. This combination improves the efficacy of antibiotics while minimizing off-target effects, thereby reducing the likelihood of adverse reactions and preserving the beneficial microbiota of the host. The typical structure of an AAC includes an antibody linked to an antibiotic via a chemical linker, which is cleaved once the complex is internalized by the bacterial cell, releasing the antibiotic to exert its bactericidal effect.
Clinical Applications
One notable example of an AAC is RG7861 (also known as DSTA4637A), developed to target Staphylococcus aureus, including methicillin-resistant S. aureus (MRSA). This AAC utilizes a human monoclonal antibody against wall teichoic acid (WTA) coupled with a rifamycin derivative. It first binds to S. aureus bacteria, and the Fc domain of the mAb is recognized by the FcRn on the surface of professional phagocytes or other host cells, such as epithelial cells. The complex is then internalized and a fusion between the phagosome and lysosome and cleavage of the VC linker occurs. The active dmDNA31 antibiotic is released attacking the intracellular bacteria, effectively killing both active and dormant bacterial populations. RG7861 has shown promising results in preclinical and early clinical trials, demonstrating its potential to treat challenging infections.2
Challenges and Future Directions
Despite their promise, the development and deployment of AACs face several challenges:
- Complexity of Development: Creating effective AACs involves complex bioengineering to ensure the stability and functionality of both the antibody and the antibiotic components. The linker technology, which must be stable in the bloodstream but cleavable in the bacterial environment, adds another layer of complexity.
- Cost: The production of AACs is more expensive than traditional antibiotics, potentially limiting their widespread use. However, as the technology matures and production methods are optimized, costs may decrease.
- Future research will likely focus on improving the efficiency of AAC production, optimizing linker technologies, and expanding the range of bacterial targets. Additionally, combining AACs with other therapeutic strategies, such as vaccines and immune modulators, could provide a multifaceted approach to combating bacterial infections and AMR.
In future perspectives, we may start to think about the use of antimicrobial peptides (AMPs) instead of just antibiotics, because of the increased resistance of pathogens (AMR). AMPs have several important characteristics, including specificity, potency, low toxicity, and biological diversity, and they can act on various targets on the plasma membrane and intracellular targets of pathogenic bacteria. They also possess antibacterial and anticancer properties, and thus is a new treatment option for cancer patients.3
- Cavaco, M., B. Castanho, A. R., & Neves, V. (2022). The Use of Antibody-Antibiotic Conjugates to Fight Bacterial Infections. Frontiers in Microbiology, 13. https://doi.org/10.3389/fmicb.2022.835677
- Peck, M., Rothenberg, M. E., Deng, R., Lewin-Koh, N., She, G., Kamath, A. V., Carrasco-Triguero, M., Saad, O., Castro, A., Teufel, L., Dickerson, D. S., Leonardelli, M., & Tavel, J. A. (2019). A Phase 1, Randomized, Single-Ascending-Dose Study To Investigate the Safety, Tolerability, and Pharmacokinetics of DSTA4637S, an Anti-Staphylococcus aureus Thiomab Antibody-Antibiotic Conjugate, in Healthy Volunteers. Antimicrobial Agents and Chemotherapy, 63(6). https://doi.org/10.1128/AAC.02588-18
- Zhang, Y., Yan, B., Meng, M., Hong, Y., Shao, G., Ma, J., Cheng, R., Liu, J., Kang, J., & Fu, Y. (2021). Antimicrobial peptides: Mechanism of action, activity and clinical potential. Military Medical Research, 8. https://doi.org/10.1186/s40779-021-00343-2