Following activation, a subset of B cells undergoes differentiation to become plasma cells. This transformation is driven by cytokines and co-stimulatory signals, such as those received from T helper cells. These signals activate specific transcription factors, like Blimp-1 and XBP-1, which orchestrate the transition into antibody-producing factories.
Morphological and Functional Changes:
The activated B cell to plasma cell transition involves dramatic changes, both in structure and function:
- Increased Endoplasmic Reticulum (ER): The ER is the site of antibody assembly. Plasma cells significantly expand their ER network to accommodate the high rate of antibody production.
- Increased Golgi Apparatus: The Golgi apparatus modifies and packages proteins for secretion. Plasma cells also enlarge their Golgi apparatus to efficiently package and export the large number of antibodies they produce.
- Reduced Ribosomes: Activated B cells possess numerous ribosomes for protein synthesis, but plasma cells drastically reduce the number of ribosomes as they shift their focus solely to antibody production.
- Abundant Rough Endoplasmic Reticulum (RER): The RER is the part of the ER involved in protein synthesis and is studded with ribosomes. While the overall number of ribosomes decreases, the proportion of RER within the ER increases in plasma cells due to its critical role in antibody production.
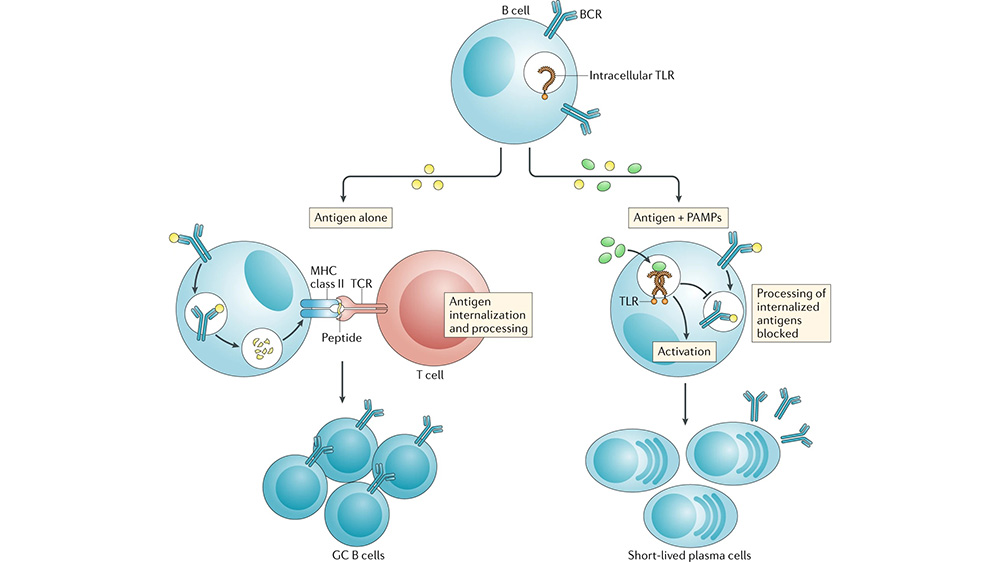
- Loss of Antigen Presentation Machinery: Activated B cells can present antigen fragments to T cells. However, plasma cells lose this capability as they focus solely on antibody production.
Functional Specialization:
- Antibody Production: Plasma cells become highly specialized factories dedicated to the production and secretion of large quantities of antibodies specifically targeted against the encountered antigen. This massive antibody production requires increased metabolic activity to fuel the cellular processes involved.
- Short Lifespan: Unlike memory B cells which can persist for years, plasma cells have a relatively short lifespan, typically lasting several days to weeks. This ensures a controlled immune response and prevents excessive antibody production.1
Following their rapid production within plasma cells, antibodies embark on a crucial journey. Inside these specialized cells, newly synthesized antibody chains undergo complex folding and assembly, aided by chaperone proteins. Additionally, sugar molecules are attached to the antibodies in the Golgi apparatus, enhancing their stability and functionality. These assembled and modified antibodies are then meticulously packaged into vesicles within the Golgi, ready for transport. Upon receiving specific signals, these vesicles fuse with the plasma cell membrane, releasing the antibodies through a process called exocytosis. The released antibodies can travel to various locations in the body depending on their type. Some circulate freely in the bloodstream, patrolling for invaders, while others accumulate in specific tissues like mucous membranes, providing localized defenses.2
Once released, antibodies play a vital role in combating pathogens. They can directly bind to and neutralize specific antigens on pathogens, rendering them inactive. Additionally, they can act as opsonins, marking pathogens for destruction by immune cells like macrophages, or even trigger the complement system, a cascade of proteins that further aids in pathogen elimination. The intricate mechanisms of antibody secretion and release thus ensure their efficient delivery and deployment, empowering them to effectively neutralize pathogens and protect the body from infections.
References:
- Cavacini, L. (2010). Structure and Function of Immunoglobulins. The Journal of Allergy and Clinical Immunology, 125(2 0 2), S41. https://doi.org/10.1016/j.jaci.2009.09.046
- Nutt, S. L., Hodgkin, P. D., Tarlinton, D. M., & Corcoran, L. M. (2015). The generation of antibody-secreting plasma cells. Nature Reviews Immunology, 15(3), 160-171. https://doi.org/10.1038/nri3795